Professor Steve Griffin
- Position
- Professor of Cancer Virology
- Areas of expertise
- Antivirals; Viroporins; Viral Oncology; Virus-mediated Immunotherapy; Liver Cancer
- Location
- Wellcome Trust Brenner Building, 5.12
- Faculty
- Medicine and Health
- School
- Leeds Institute of Molecular Research, School of Medicine
Introduction
Our laboratory focuses upon two important aspects of human virology. The first is the study of emerging viruses with pandemic potential and the development of new antiviral drugs. The second is the role played by viruses as both causative, and potentially curative, agents in human cancers, with a particular focus on primary malignancies of the liver.
Our key areas of interest are:
- The study of emerging or global viruses including hepatitis C virus (HCV), pandemic influenza A virus (IAV), Zika virus (ZIKV), and severe acute respiratory syndrome coronavirus type 2 (SARS-CoV2), the aetiological agent of COVID-19. In particular, we aim to understand and develop new antiviral therapies targeting virus-encoded ion channels, a.k.a. “viroporins”.
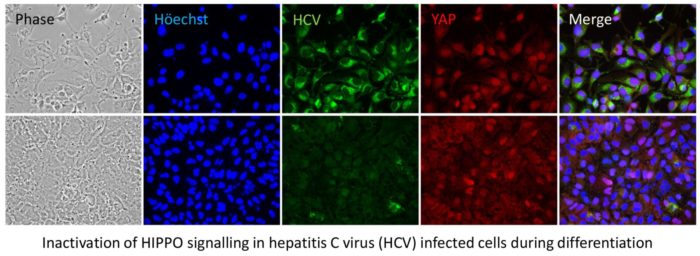
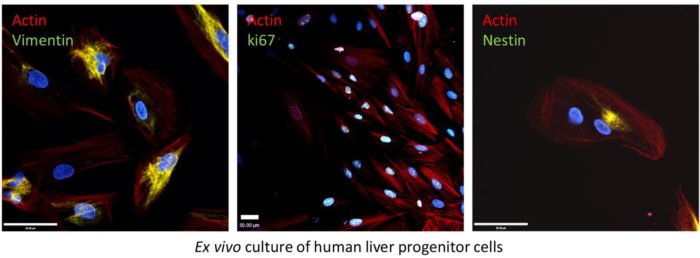
- Understanding how HCV and human cytomegalovirus (hCMV) manipulate tissue progenitor cells as a potential cause of human malignancy. For HCV we focus on primary liver tumours (hepato- or intrahepatic cholangio- carcinomas, as well as hepatoblastoma), whereas hCMV is studied in relation to brain tumours (glioblastoma multiforme).
- Investigating the immunological processes underpinning virus-mediated immunotherapy for human liver cancers, both from viral and non-viral origins. We focus upon a unique strategy comprising modified “oncolytic” viruses combined with tumour conditioning using tyrosine kinase inhibitory drugs.
Current major projects
- Antiviral therapies for emerging and global viral pathogens.
- Viral oncology
- Virus-mediated immunotherapy for liver cancer
Detailed research programme
Antiviral therapies for emerging and global viral pathogens
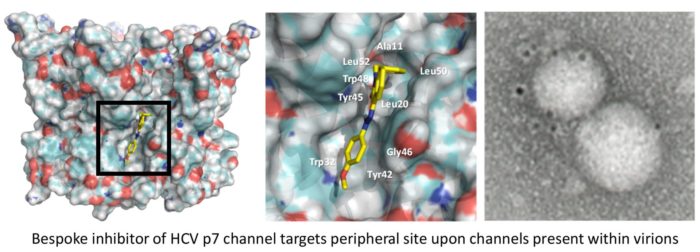
Since discovering the ion channel function of the HCV p7 protein, we have been interested in viroporins encoded by viruses with significant impact upon human health. With our collaborators, we adopt a rational medicinal chemistry approach, informed by structural biology and molecular dynamics, to identify druggable targets and develop potent and specific small molecules. In addition to their therapeutic applications, inhibitors also serve as excellent molecular tools with which to understand viroporin function in the life cycle of these important human pathogens.
Viral Oncology
Since the advent of new direct-acting antiviral therapies, one of the major clinical problems remaining associated with HCV infection is the major role it plays in the development of liver cancers. Moreover, it appears that this risk persists after patients have been “cured”. We are interested in how HCV infection changes the behaviour of liver progenitor cells, potentially priming them to become malignant “cancer initiating cells” that drive both hepatocellular, and intra-hepatic cholangio- carcinomas. In addition, we are also exploring the controversial association of hCMV with brain tumours, specifically glioblastoma multiforme, which also appears to involve progenitor cells in the adult brain.
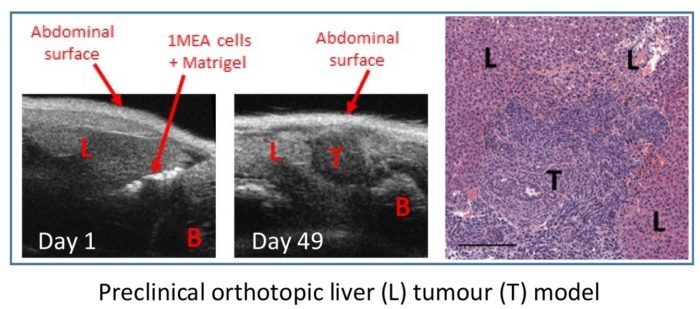
Virus-mediated immunotherapy for liver cancers
Immunotherapy is becoming a mainstay for the treatment of solid tumours, achieving remarkable results in otherwise incurable disease. However, many patients still do not respond to the immune checkpoint antibodies widely employed today. Another form of immunotherapy can be achieved using so-called “oncolytic viruses” (OV), which act as potent adjuvants to the immune system.
We are investigating the immunological mechanisms underpinning a remarkable therapeutic response achieved within models of liver cancer, using a combination of a modified OV and manipulation of tyrosine kinase signalling. We hope to reveal key responses capable of destroying liver tumours, and to improve this regimen as a future therapeutic.