Dr Paul Beales
- Position
- Professor of Soft Matter and Biophysics
- Areas of expertise
- Membrane biophysics; Bionanotechnology; Artificial Cells; Nanomedicine; Hybrid lipid-polymer vesicles
- Phone
- +44(0) 113 343 9101
- Location
- 1.13 Chemistry Building
- School
- Chemistry

Introduction
Membranes define the interface between the cell and its environment, compartmentalise processes and control transport of matter and signals between these compartments. We are interested in understanding the biophysical properties of minimal model membranes to provide new insights to biological mechanisms and processes. Our approach is to apply physical sciences concepts from the field of soft matter to improve fundamental understanding of these systems. Furthermore, we take inspiration from biological membranes and compartmentalisation to develop new materials for biotechnology and medicine. One long-term ambition of my group is the bottom-up assembly of artificial cells: minimal functional compartments inspired by the function of natural cells.
Current major projects
- Development and mechanistic understanding of membrane-active anticancer peptides
- Material properties of hybrid lipid-block copolymer membranes and their application to membrane protein biotechnology and nanomedicine
- Reconstitution and functional mechanisms of ESCRT membrane remodelling complexes and their application in fabrication of compartmentalised artificial cells
- Confinement of feedback-responsive enzyme reactions: mechanisms and applications
Detailed research programme
Understanding and developing anticancer peptides
The membranes of cancer cells undergo pathological changes in the lipid composition. Similar to how antibiotic peptides of the innate immune system target differences in the membrane composition of bacterial cells compared to native eukaryotic membranes, some pore-forming peptides are known to be more potent against cancer cells. We are using a range of approaches from model membranes to cells to understand the mechanism of action of these peptides and seek to enhance their potency and specificity to cancer cells towards potential therapeutic applications.

Hybrid vesicles for membrane protein stabilisation
Hybrid vesicles composed of lipids and block copolymers can combine the advantages of native-like lipid membranes with the enhanced stability and broader chemical parameter space of block copolymer membranes. We have developed techniques to reconstitute integral membrane proteins into hybrid vesicles and demonstrated that this can significantly enhance the in vitro functional durability of the membrane protein. We are interested in developing this approach for a range of applications including biofuel cells, pharmaceutical assays, biocatalysis and artificial cells.
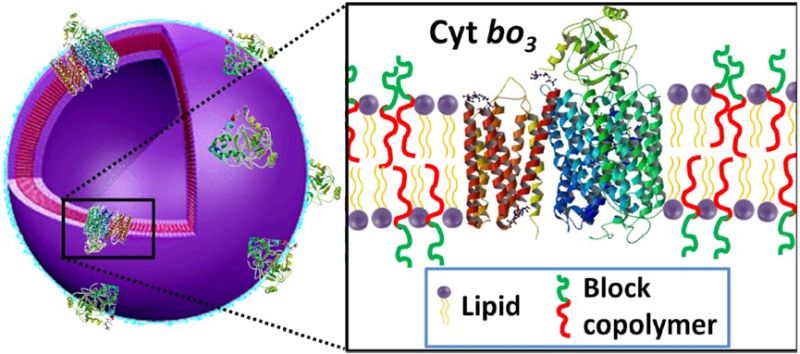
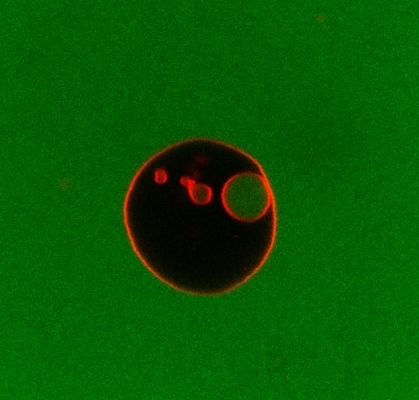
Bottom-up assembly of artificial cells
There is currently significant interest in the assembly of cell-like materials from their molecular constituents. These compartmentalised systems take inspiration from biological cells through incorporation of functions like encapsulated metabolisms, communication with and response to their environment. We are contributing to development of the synthetic biology toolbox for designing artificial cells for practical applications. Examples include repurposing ESCRT membrane remodelling proteins to fabricate multicompartment eukaryote-like cell architectures and encapsulation of feedback-controlled enzymatic reactions for responsive and adaptive function.
Soft materials for medical technologies
Inspired by biological matter and applying concepts from soft matter, we are interested in developing new materials for applications in medicine. This includes vesicle nanomedicines for controlled and targeted delivery of therapeutics in the body and injectable hydrogels for tissue regeneration.