Professor Dejian Zhou
- Position
- Professor of Nanochemistry
- Areas of expertise
- Biomedical Nanomaterials; Biosensing; Multivalent protein-sugar interaction; Cancer nanomedicine; nano-antibiotics
- d.zhou@leeds.ac.uk
- Phone
- +44(0)113 343 6230
- Location
- 1.54 Chemistry
- Faculty
- Engineering and Physical Sciences
- School
- Chemistry
Introduction
We are developing a novel polyvalent multifunctional nanoparticle (PMN) strategy to exploit multivalency (e.g. greatly enhanced binding affinity and specificity) and further integrate the unique, size-/shape- dependent optical and magnetic properties of functional nanoparticles to address some important, unmet biological and biomedical challenges. Our current research interests include: glycan-PMNs as novel multifunctional tools to unravel the structural, molecular and cellular mechanisms of multivalent lectin-glycan interactions underlying the HIV/Ebola infections and to exploit such new understanding to develop novel immunotherapeutic strategies; PMN-ultrasensitive assays for early, non-invasive detection and diagnosis of cancer and other important human diseases; PMN-cancer nanomedicine for effective, targeted multimodal imaging and treatment of cancer to address tumour multidrug resistance; and PMN-antibiotics to overcome bacterial antibiotic resistance.
Current major projects
- Glycan-PMN mechanistic probes for virus-dendritic cell interaction and therapeutic interventions
- PMN ultrasensitive assays for early, non-invasive disease diagnostics
- PMN cancer nanomedicine for effective, targeted multimodal cancer theranostics
- PMN anti-bacterial agents for addressing antibiotic resistance
Detailed research programme
Glycan-PMN Probes
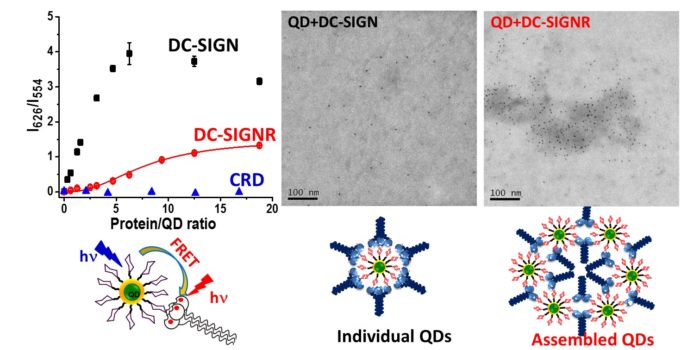
Multivalent lectin-glycan interactions are central to viral infection and regulation of immune response, but the structural mechanisms remain poorly understood, making it difficult to develop glycoconjugate therapeutic interventions. We have pioneered the use of glycan-PMNs as virus mimics to probe virus-dendritic cell (DC, via its surface tetrameric lectin DC-SIGN) interactions. The glycan-PMNs can not only quantify binding affinity, but also dissect the exact binding mode and potently block DC-SIGN mediated Ebola virus infection of host cells whose potency is directly linked to its DC-SIGN binding affinity/mode. The glycan-PMNs are being exploited as multifunctional probes for DC immune regulation mechanisms and novel immunotherapeutic agents against important diseases such as allergy and arthritis.
PMN Ultrasensitive Assays
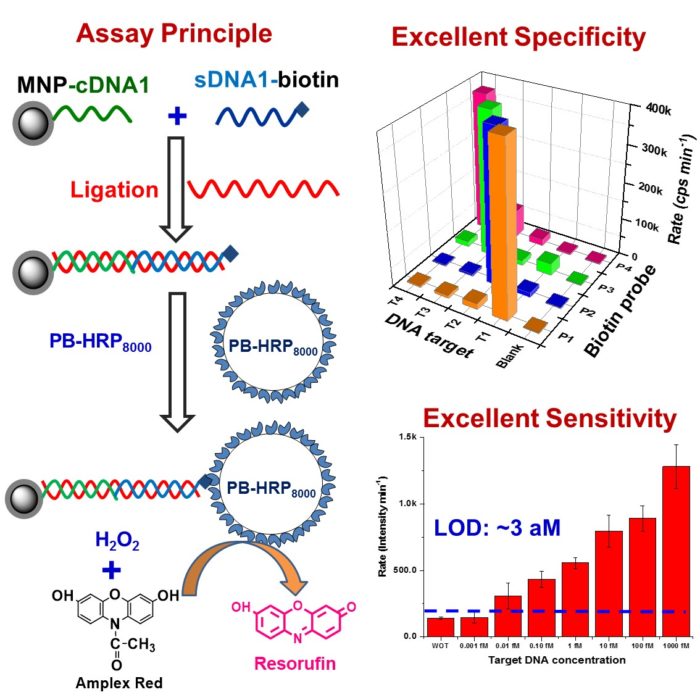
The clinical "gold standard” assay typically detect target proteins down to the pM level (10-12 M), limiting its capability in early disease diagnosis where the biomarker concentrations can be 3 orders of magnitude lower. By exploiting the unique super-paramagnetic properties of magnetic nanoparticles (MNPs) and the great signal amplification power of poly-enzyme tagged polymer nanobeads, we are developing ultrasensitive PMN sensors that can directly and specifically detect target biomarkers down to the low atto-molar (10-18 M) regime, making them powerful tools for non-invasive, earlier detection and diagnosis of important diseases such as cancer. We are further exploiting its applications in clinical diagnostics.
PMN Cancer Nanomedicine
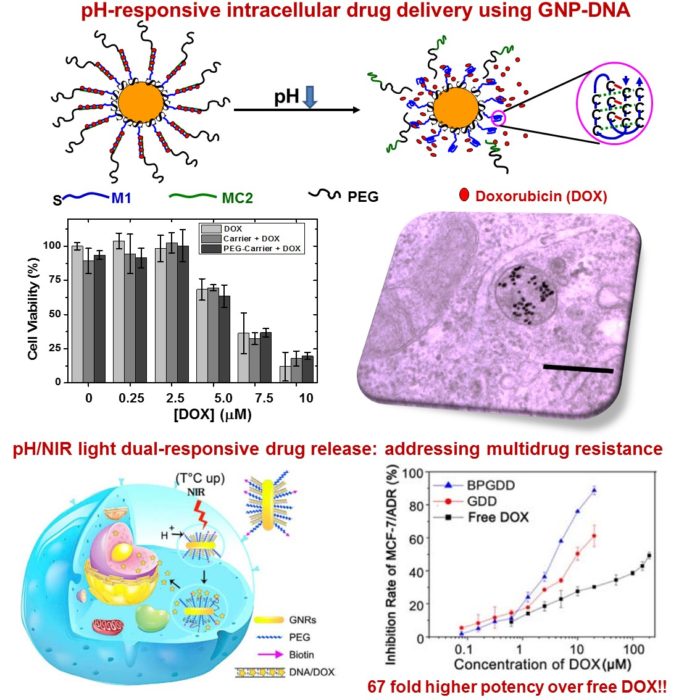
Cancer is a leading cause of death worldwide, accounting for 9.6 million deaths and >US$1.16 trillion annual economic cost. The current treatments are ineffective in treating metastatic and multidrug resistance cancers. To address this challenge, we are developing PMN nanomedicine to exploiting multivalent binding between PMN surface targeting ligands and cancer cell over-expressed receptors and tumour’s own pathological conditions (e.g. enhanced permeation and retention, EPR) effect to improve targeted tumour accumulation. Meanwhile, we are integrating the unique size-/shape- dependent physical-/chemical- properties of nanoparticles to offer stimuli-response release, imaging, diagnostic, and therapeutic functions. We are further incorporating chemotherapeutic and immune modulating functions to offer more effective, synergistic multimodal treatment to overcome cancer multidrug resistance at the cellular and pre-clinical levels.
PMN Antibiotics
The emergence of resistant bacteria (e.g. MSRA, VRE) has been a major global health problem. Previously, in collaboration with Prof. Rachel McKendry (University College London) we have developed a microcantilever array based rapid screening method for vancomycin-mucopeptide interactions (Nature Nanotech. 2008 and BBC highlight). We are developing novel PMN antibiotics to enhance the potency of existing antibiotics by exploiting multivalency and harnessing intrinsic anti-bacterial properties of nanomaterials. As a result, the PMN antibiotics can offer a safer, faster, and more economical alternative to traditional medicinal chemistry approaches to combat the bacterial antibiotic resistance challenge (in collaboration with Dr Alex O’Neill, Faculty of Biological Sciences, Leeds and Prof. Xingyu Jing, Southern University of Science and Technology, China).