Professor Adam Nelson (Deputy Director)
- Position
- Professor of Chemical Biology
- Areas of expertise
- chemical biology; activity-directed synthesis; chemical space; bioactive molecular discovery; chemical tools
- Phone
- +44(0)113 343 6502
- Location
- G.30 Chemistry
- Faculty
- Engineering and Physical Sciences
- School
- Chemistry
Introduction
The discovery of bioactive small molecules is an enduring challenge in both chemical biology and medicinal chemistry. Small-molecule drugs dominate our collective ability to treat disease, and the availability of high-quality chemical probes can help address the uneven investigation of the biology of proteins. However, the chemical reaction toolkit that underpins bioactive molecular discovery is remarkably narrow which has contributed to the uneven historical exploration of chemical space. My group has pioneered innovative chemical approaches to drive the discovery of functional small molecules, and to unlock new biologically-relevant regions of chemical space. We harness these approaches to discovery novel chemical tools that enable the investigation of fundamental biological mechanisms.
Current major projects
- Activity-directed synthesis of bioactive small molecules
- Fragment-oriented synthesis
Detailed research programme
Activity-directed synthesis of bioactive small molecules
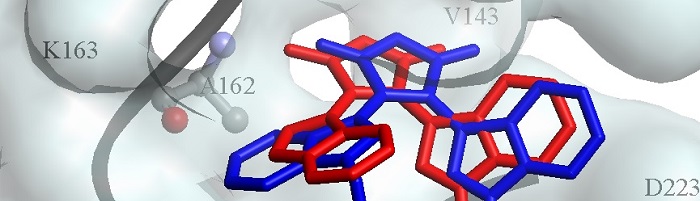
Human-driven approaches to bioactive molecular discovery are generally based on iterative cycles of molecule design, synthesis, purification and evaluation. In stark contrast, the evolution of biosynthetic pathways to bioactive natural products is structure-blind and function driven. The diverse structures of natural products have inspired the discovery of many bioactive small molecules including around 35% of small-molecule drugs. We are developing a novel discovery approach, activity-directed synthesis, which has some parallels with the evolution of biosynthetic pathways. The approach intentionally harnesses promiscuous chemical reactions to explore novel chemical space, and can enable the discovery of bioactive molecules with unexpected structures as well as an associated synthesis. We are exploiting activity-directed synthesis in the discovery of many classes of ligand including protein-protein interaction inhibitors, antibacterials, antiparasitic compounds, and protein kinases.
Fragment-oriented synthesis
Fragment-based discovery approaches are now widely applied for the discovery of novel bioactive small molecules. Here, fragment sets are initially screened against a target protein, and hit fragments typically bind weakly, yet efficiently. Chemical space may be explored efficiently because the number of possible compounds scales exponentially with molecular size. Fragment hits are elaborated to yield higher affinity ligands. Unfortunately, fragments are rarely elaborated directly: instead, de novo synthesis of “grown” fragments is usually required. We are developing fragment-oriented synthesis in which fragment hits, particularly three-dimensional fragments, may be elaborated directly along many alternative vectors. We are exploiting the approach in the discovery ligands for a wide range of targets including protein-protein interactions and protein kinases.